With the pervasiveness of e-Commerce for the sale of non-prescription medicines, there has been a growing global concern, in recent years, about the need to establish new and unique regulations to enhance the fight against the distribution of counterfeit medicines. In the European context, Directive 2011/62/EU was introduced to hinder the illegal trade of medicines and amend the previous Directive 2001/83/EC, which established a Community code for medicinal products for human use. With this important update, a series of measures were brought about to prevent and combat the problem of falsified medicines through the serialisation process of pharmaceutical products.
Packaging serialisation: a key element for drug traceability
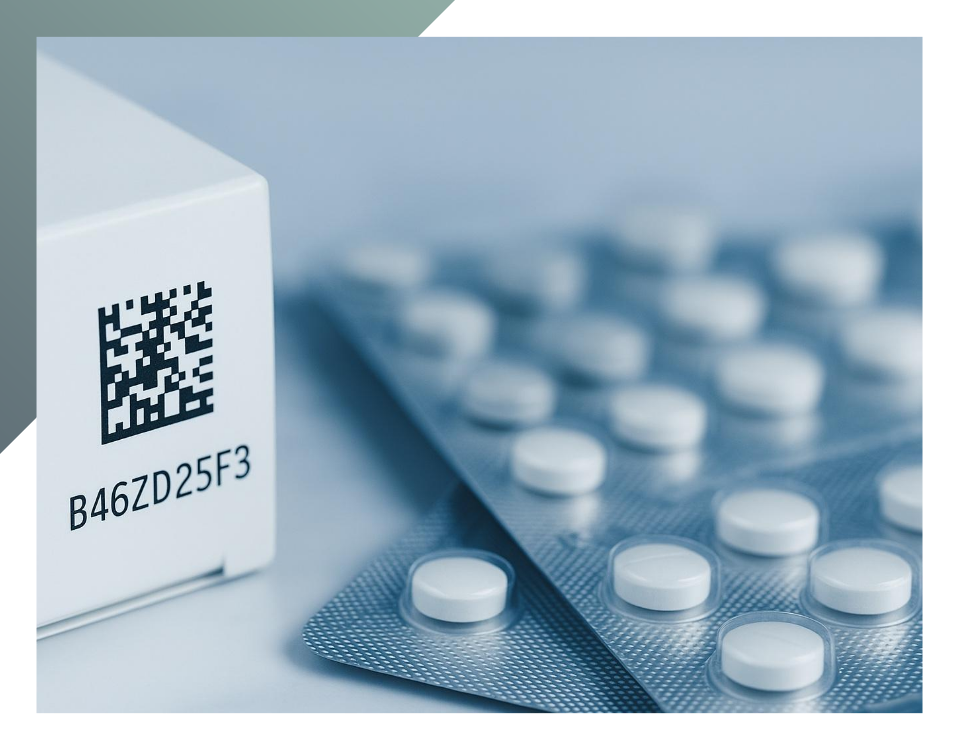
The serialisation of medicines is the mandatory adoption of new coding systems applied on the packaging, globally valid, to reduce the introduction of counterfeit pharmaceuticals that represent a dangerous threat to public health because they may contain low-quality and toxic ingredients in the wrong dosages or lack essential active ingredients. However, to date, not all countries have yet adopted this system. Among the countries that will adopt the new coding solutions, there is also Italy, which already has its an efficient traceability system, which we will discuss in more detail later in this article. Directive 2011/62/EU – “Falsified Medicines Directive” (FMD) of the European Parliament and the Council defines a falsified medicinal product i.e «any medicinal product with a false representation of»:
- Identity (packaging, labelling, name or composition, concerning any of the constituents, including excipients and dosage);
- Source, including manufacturer, country of manufacture, MA (marketing authorisation) holder;
- Logistical and documentary history, including records of distribution channels, was involved.
This last point highlights how drug traceability is a common element in all initiatives to combat counterfeit medicines and to ensure to combating counterfeit pharmaceutical and ensuring the safety of patients who consume them. It is a crucial element in international pharmaceutical supply chains, it can be guaranteed only through the serialization of drugs and the assignment of a predetermined code (the Data Matrix code) to the different packaging levels of a product: multi-pack units, outer packaging, and pallets. This code may vary depending on the specific sector. Generally, when encoded, it contains the following information:
- Global Trade Identification Number (GTIN) which identifies the product or manufacturer
- The unique serialisation number;
- Additional codes.
Information such as the lot number, date of manufacture and expiry date of the drug are usually printed in plain text on the packaging. This system allows monitoring of the path of the products from the production plant through all logistics operations until they reach the final sales channels (intended for individual consumers or hospitals). A secure and effective traceability system can uniquely identify each package at every level of the supply chain, immediately determining its origin and movements.
The Italian traceability system before the European Directive
Italy has long been at the forefront of implementing a comprehensive drug-tracking system throughout its distribution chain. Before the introduction of Directive 2011/62/EU, the Italian pharmacovigilance system already had its advanced drug tracing protocol based on anti-counterfeiting labels produced by the Institute of the Italian State Mint and Polygraphic Institute and applied to pharmaceutical packaging. The tags are adhesive containing a barcode with all the necessary information to identify the medicine and its package, including the following elements:
- Holder and drug name;
- Marketing authorisation number (MA);
- Unique sequential number of the label itself.
The tag can be scanned at any point in the distribution chain and communicated to a central database managed by the Italian Ministry of Health, which keeps all production data and progressive label numbers associated with each package of medicines in the market. In addition to ensuring the authenticity of medicines, this tracking system allows the Ministry of Health to verify the type and number of prescribed drugs in the territory and obtain some useful data such as the demographic characteristics of the patients they are intended for (prevalence of certain diseases, geographical areas with higher prescription rates, and for which therapies they are used).
An innovation introduced by the EU Directive: the Data Matrix code
With a few exceptions as China, which uses its linear 1D serialization system, the most countries in the world have already implemented the Data Matrix code. For EU Member States, the obligation to apply the Data Matrix according to GS1 standards on every package was introduced as on 1st January 2016. The Delegated Regulation (EU) 2016/16 sets the safety requirements for the packaging of human medicines, it regulates all necessary standards for ensuring the conformity of the Data Matrix code, in its structure and printing on the packaging (such as the contrast between light and dark parts or legibility). Due to the presence of its existing pharmaceutical tracking system represented by the anti-counterfeiting label, Italy has been granted an exemption until 2025 for the introduction of the Data Matrix code. This system will serve as an additional tool to support the traceability of medicines from the early stages of procurement to the final sale. Likewise Italian anti-counterfeiting tag, Data Matrix codes are directly linked to a database containing information about registered medicines at the time of production. This hub is the European Medicines Verification System (EMVS), managed by the European Medicines Verification Organization (EMVO). When each Data Matrix code is assigned, the data is sent to the European hub, which sends it to the national database of the relevant country, known as the National Medicines Verification Organization (NMVS), for registration. The code can be scanned at any stage of the distribution chain, allowing for a comparison between the information registered in the central European hub and the databases of each nation. Adopting Data Matrix codes and a unified European monitoring system brings several advantages in optimizing anti-counterfeiting protocols. Data Matrix code can be checked easily through automated operations and allows the encoding a large amount of information that could not be contained in a traditional barcode. In addition to the Data Matrix, European legislation requires manufacturers to include an Anti-Tampering Device (ATD) to ensure the integrity of the packaging. If the package is opened it cannot be resealed in the same way, thus indicating any potential tampering.
Intelligent labels supporting the serialization process: RFID and NFC tags
In addition to well-established reading tools, the pharmaceutical sector is increasingly adopting technological packaging solutions to support fast and secure encoding of the Data Matrix, starting from the pallet tracking phase. RFID (Radio Frequency Identification) technology, for example, is a radio-frequency-based communication method that uses special tags (electronic labels with a memory chip inside) to transmit the Data Matrix code and associated information at varying distances. Rather than scanning individual packages, RFID technology allows the scanning of codes as pallets pass through dedicated gates in the distribution chain. The information stored in the memory chip, cannot be modified because each tag has a unique Transponder Identification (TID) code known only to the label manufacturer. NFC (Near Field Communication) tags, to be considered the evolution of RFID technology, work similarly. They are electronic tags scannable on time when brought close to a compatible device, including smartphones. They provide access to a range of consumer-oriented information about the product and its entire distribution chain, including traceability certifications. The Eurpack Packaging Development Center is responsible for developing and customizing solutions that meet international security standards according to the specific request of each customer.
How can we help you?
If you would like to learn more about our commitment to sustainability or have any questions about our projects and initiatives, we are here to provide you with all the information you need.